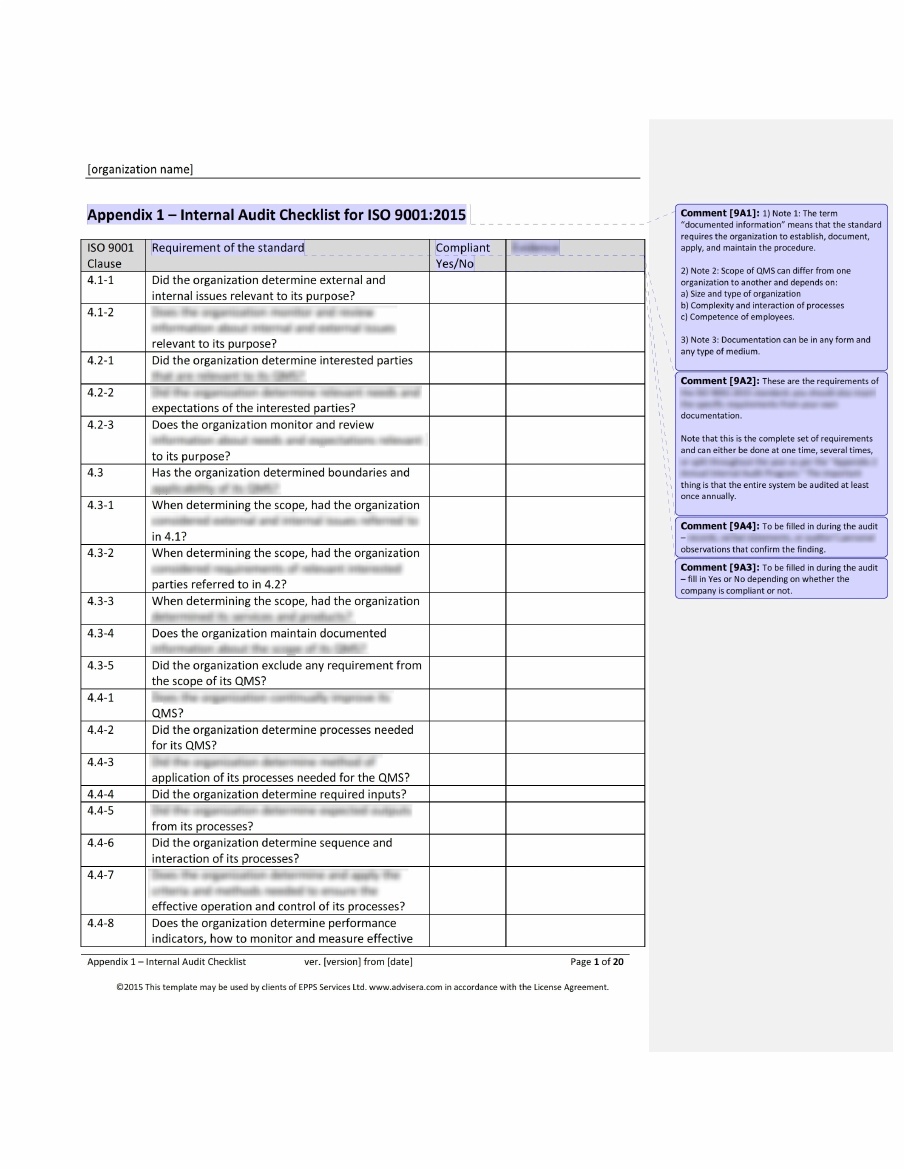
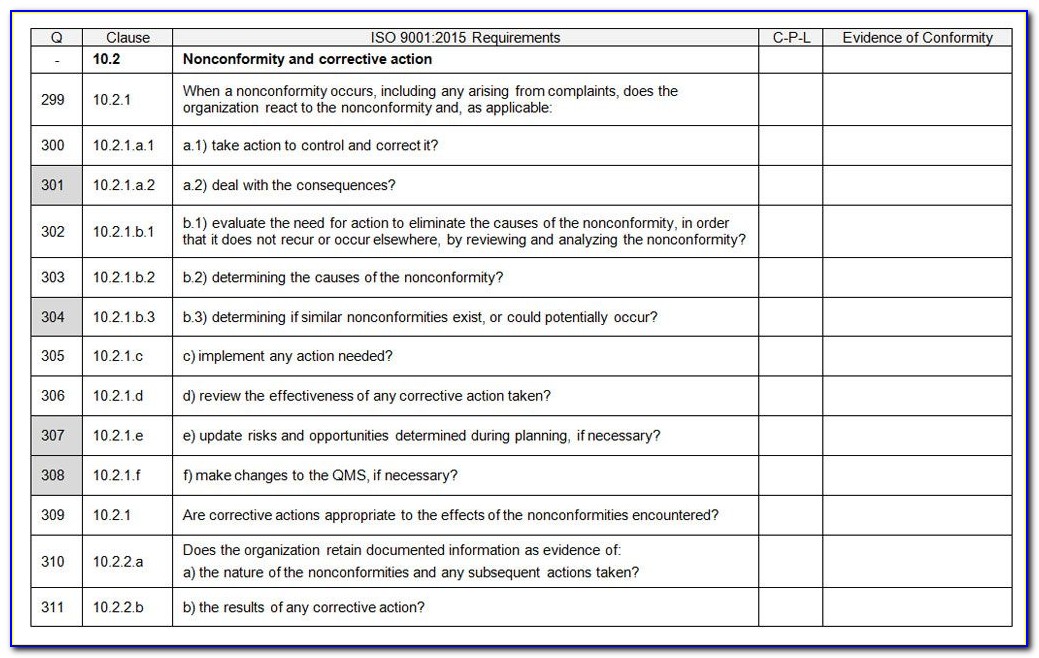
Members of the applying organization frequently execute this task to assist the complete company in preparing for the certification audit. Internal Audit: This is used to test initial compliance.This necessitates comprehensive policies, processes, work instructions, records, and other documents. Documentation: The applicant team must document how their QMS meets the standard.This need support from the entire organization. This often needs a significant cultural transformation within the firm. Preparation: The applicant must create and implement a QMS that meets the standards.The ISO 13485 auditing and accreditation process typically includes the following steps: ISO 13485’s Audit and Certification Process Management Review: Auditors examine top management’s internal QMS evaluation and improvement strategies.The organization’s process for addressing these issues may also be evaluated. The non-compliant components will then have to be re-audited. Noncompliance and Corrective Actions: Any audit deficiencies must be identified and resolved.Regulatory Conformity: The audit certifies conformity with regulatory requirements, such as those set by the FDA in the United States or the Medical Devices Directive in the European Union.
This comprises all stages, including design and development, risk management methods, supplier management techniques, and other relevant processes.

To acquire ISO 13485 certification, an organization must establish an internal QMS process that meets the standard. This includes process control from design and development to manufacture, installation, maintenance, and end-of-life. ISO 13485 certification demonstrates that the certified company has implemented appropriate, independently inspected processes and controls to assure the safety, efficacy, and quality of medical devices throughout their lifecycle. The standard prioritises regulatory compliance, risk management, and process control for medical device design, production, and distribution. ISO 13485 is based on the ISO 9001 standard however, it adds some sector-specific features. It specialises in the design, development, production, installation, and maintenance of medical devices and related services. "About this title" may belong to another edition of this title.ISO 13485 is a globally recognised standard that defines the standards for a quality management system (QMS) in the medical device industry. As an official consultant for the European Union he assists governments in Asia and Eastern Europe in setting up their ISO 9000 accreditation schemes he is a public speaker and contributor of articles at conferences and professional publications and he is an active auditor collaborating with major international registrars.

He is a prominent figure in the world of quality and environmental management systems. Kanholm's experience with auditing and certifying management systems dates back to the first European ISO 9000 certifications in the mid-eighties. He is a Registered Lead Assessor licensed by the International Register of Certified Auditors (IRCA), a Lead Auditor under the Registrar Accreditation Board (RAB) Assessor Registration Scheme, and an AIAG certified Lead Auditor.
Jack Kanholm holds an Engineering Master of Science degree from the Technical University of Denmark.
